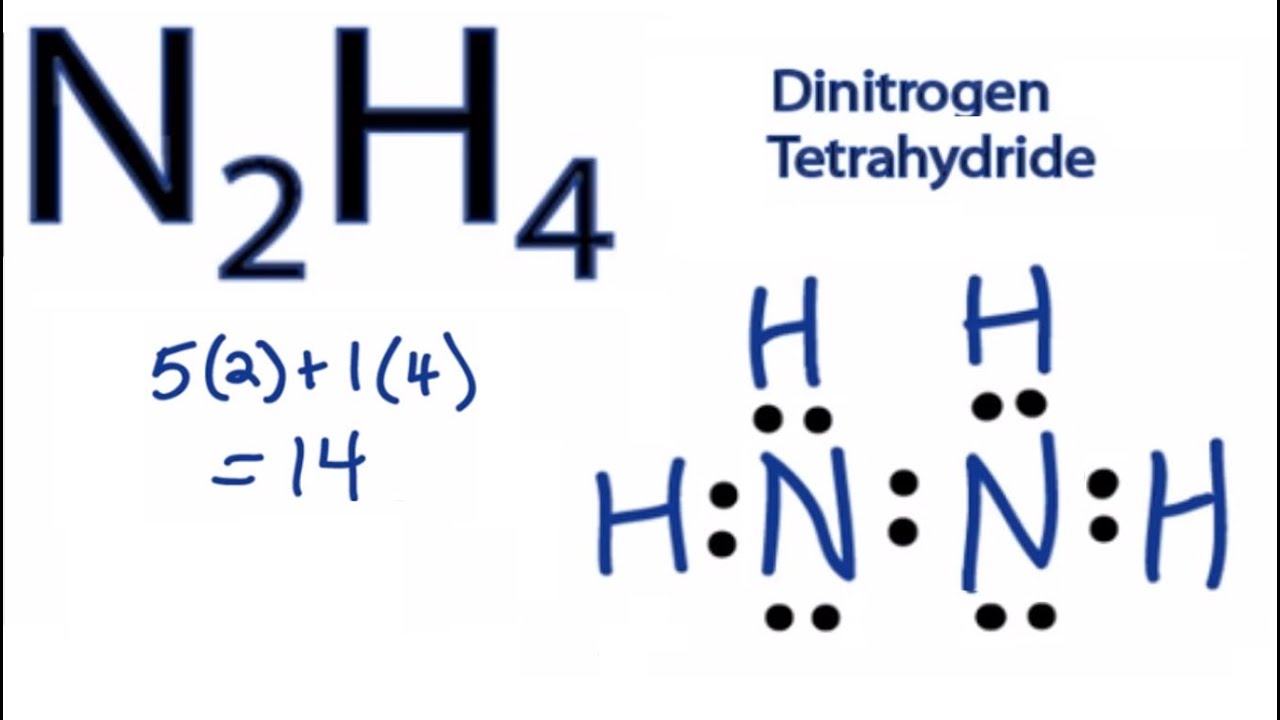
H4n2 Lewis Structure. Your friend places a single bond between the central atoms and a lone pair of electrons on each. Counting valence electrons yields eight total six from oxygen one each from the two hydrogens. As of 2015 the world hydrazine hydrate market amounted to 350 million. Each home pairs here Frenchy H two c n h.
Its supposed to have a total of 18 electrons in the lewis dot structure it does have 18. How to write a compounds Lewis Structure 1 Write correct skeletal structure for molecule more electronegative elements in terminal position and less electronegative elements in center. 3102011 This makes sense but when I try to convert it to a 3D structure. In the Lewis structure for N 2 H 4 there are a total of 14 valence electrons. Your friend places a single bond between the central atoms and a lone pair of electrons on each. 2 Calculate the total number of electrons for the Lewis structure by summing the valence electrons of each atom in the molecule.
Its supposed to have a total of 18 electrons in the lewis dot structure it does have 18.
This is because it has six valence electrons plus two more that it got from Mg. In both cases at least one nitrogen has only 6 valence electrons. Consider the Lewis structure for sulfur tetrafluoride SF 4 which contains 34 valence electrons. There are four covalent bonds in the skeleton structure for SF 4. Hydrazine is highly toxic unless handled in solution as eg hydrazine hydrate. A step-by-step explanation of how to draw the H2 Lewis Dot Structure Hydrogen gasFor the H2 structure use the periodic table to find the total number of v.